Epidemiology & Biostatistics
For more than 20 years, our experts in epidemiology and biostatistics have worked with prominent academics, clinicians, and thought leaders from leading universities and research institutions. Together, we develop compelling results for our clients in the pharmaceutical, biotechnology, medical device, and health insurance industries.
Our analyses use epidemiological and biostatistical methods to develop empirical and real-world evidence based on a range of data sources. This work has been used to support clients in many health care-related litigation matters, including those at the liability, class certification, and damages phases. We provide and support expert analysis in matters related to intellectual property, product liability, drug safety, causation assessment and causal inferences, false claims and kickbacks allegations, and securities fraud.
Clinical Trials:
- Efficacy and safety analysis of phase I-IV clinical trials
- Post hoc analyses of clinical trials data to address emergent safety issues and secondary endpoints
- Indirect product comparisons across clinical trials when head-to-head trials are not available
- Meta-analysis and network meta-analysis of pooled clinical trial data
- Personalized or precision medicine analysis to identify high-value subgroups
Observational Studies:
- Descriptive prevalence and incidence studies
- Pharmacovigilance and spontaneous report adverse event analyses
- Post-marketing drug safety epidemiology studies
- Registries and active surveillance
- Quality of life and patient-reported outcome studies
- Risk-benefit analysis
- Comparative studies to evaluate causality between treatments and efficacy and safety outcomes using real-world practice data
- Analysis of a drug’s patent and the potential making and selling of a generic prior to the patent’s expiration
- Signal detection in social media data
- Machine learning and digital health technologies
-
Health Care Bulletin
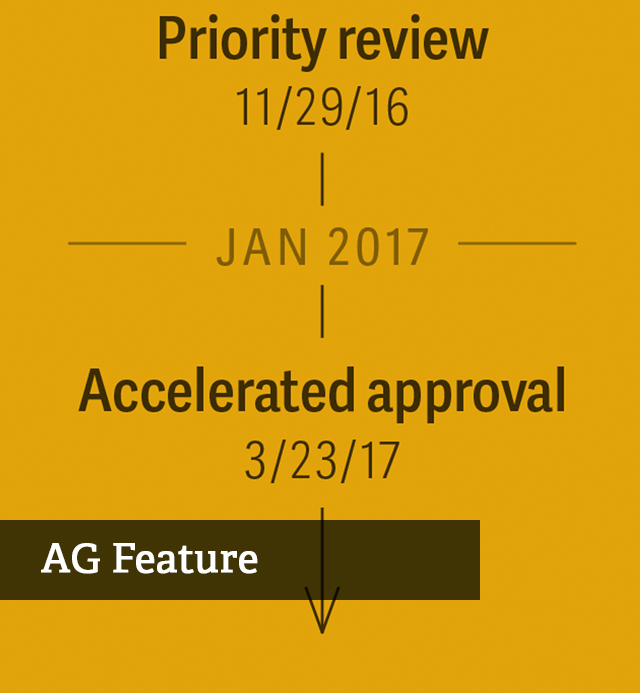
An Expanded Role for Real-World Evidence in FDA Approvals for Drug Registration
-
Featured Expert
Mei Sheng Duh
Managing Principal, Boston

- Related Focus Area Drug Safety Litigation
-
Featured Expert
Pierre Y. Cremieux
President, Boston, Paris, Brussels, London, Montreal

-
Publication
Health Care Bulletin 2021

-
Featured Expert
Daniel O. Scharfstein
Professor and Chief, Division of Biostatistics; Professor, Department of Population Health Sciences; and Adjunct Professor, Department of Orthopaedics and Department of Family and Preventive Medicine, University of Utah School of Medicine

-
AG Feature
Causation in Product Liability: Taking a New Look
-
Forum
Investigating Adverse Effects Using Epidemiology

-
Featured Expert
Lee-Jen Wei
Professor of Biostatistics, Harvard T.H. Chan School of Public Health



